New drugs against
Alzheimer’s diseasebased on monoclonal antibodies, gain on average around 10 months of autonomy, at the cost of heavy infusions once or twice a month. For patients and their loved ones, this rhythm is a constant reminder of the progression of the disease.
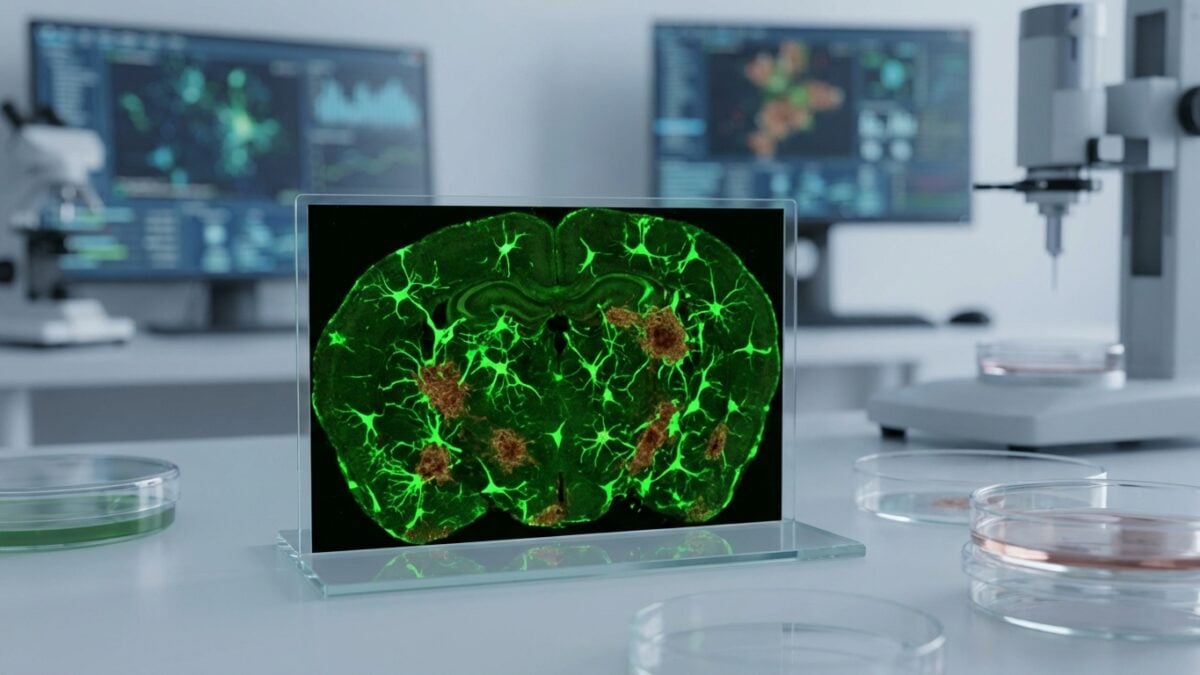
At the heart of Alzheimer’s, a sticky protein, amyloid beta, builds up in plaques in the brain and eventually derails memory. The microglia, responsible for cleaning up this waste, quickly find themselves overwhelmed. A team from Washington University in St. Louis therefore bet on a bold idea: transforming other brain cells into enhanced brain cellscapable of tracking down these toxic masses.
Astrocytes transformed into super brain cleaners
Current antibody treatments also bind to amyloid, but they remain in the blood and have difficulty crossing the barrier that protects the brain. They require high and repeated doses, with a risk of side effects visible on imaging, which limits their room for maneuver.
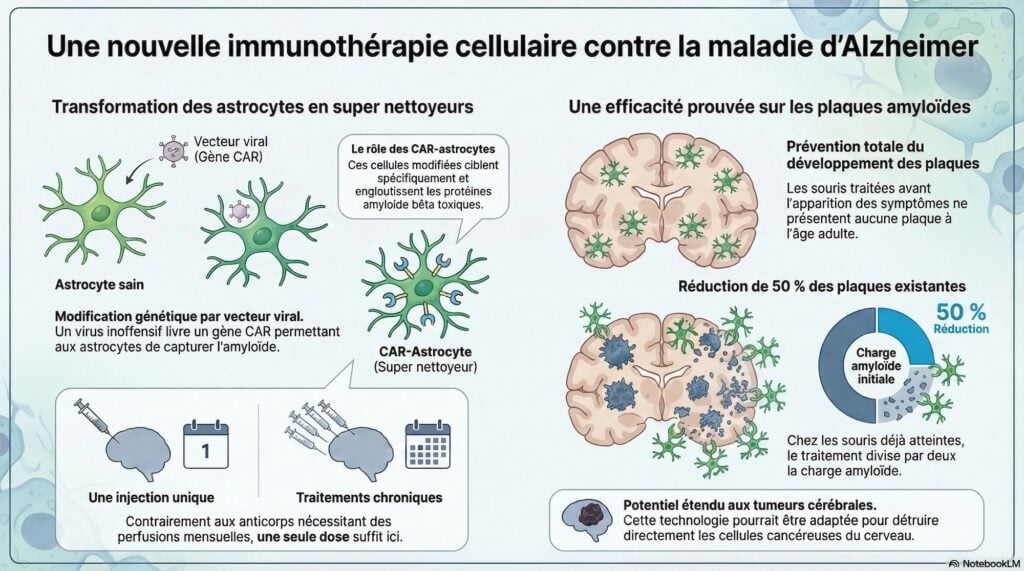
Researchers at Washington University in St. Louis have developed a innovative cellular immunotherapy to fight Alzheimer’s disease. Inspired by cancer treatments, they genetically modified astrocytesthe most numerous brain cells in the brain, to transform them into cleaning agents capable of eliminating
amyloid plaques. They added a chimeric antigen receptor, or CAR, a sort of “guidance device” that specifically recognizes amyloid beta proteins. Injected via a harmless virus, this gene transforms astrocytes into CAR-astrocytesreal “super cleaners”.
“This study marks the first successful attempt to engineer astrocytes to specifically target and eliminate beta-amyloid plaques in the brains of mice with Alzheimer’s disease. said lead study author Dr. Marco Colonna, professor of pathology at WashU Medicine.
A single injection that blocks or reduces plaques in mice
The team used mice carrying mutations which cause, around 6 months, a brain saturated with amyloid-beta plaques. Two groups received the virus carrying the CAR gene: young people, before the plaques appeared, and older groups, already invaded. Three months later, the brains treated upstream were almost free of plaques, while the controls were full of them.
In already affected animals, a single injection reduced the amount of plaques by around 50% compared to mice receiving a virus without the CAR gene. The analyzes also indicate a reduction in neuronal dystrophy and better preservation of synaptic connections and certain memory performance. The authors describe astrocytes focused on cleaning and microglia returning to a more “rested” state.
 © NotebookLM
© NotebookLM
Unlike current antibody treatments which require frequent injections, this approach requires only one single intervention to act sustainably.
What avenues before humans and beyond Alzheimer’s
The signals remain encouraging in mice, without marked neuroinflammation or damage to the blood-brain barrier. But caution prevails. “Although further research is needed to optimize this approach and manage potential side effects, these results open a new and promising perspective for the development of astrocyte CARs in immunotherapy against neurodegenerative diseases and even brain tumors.” also indicated Dr. Marco Colonna. Studies on duration of action, long-term safety and on models closer to humans remain necessary.
The authors point out that this type of approach seems particularly powerful at the start of the disease. “Like antibody treatments, this new CAR-astrocytic immunotherapy is most effective when administered in the early stages of the disease.” said David Holtzman, professor emeritus of neurology at Wash U Medicine and co-author of the paper. “But where it stands out, and where it could have a significant impact in clinical practice, is through the single injection that reduced the amount of harmful brain proteins in mice.”.
By simply changing the target of the CAR, the team is already considering directing these modified astrocytes toward brain tumors or other diseases linked to abnormal proteins, such as certain forms of Parkinson’s or frontotemporal dementia.