Refractory cancer cells: a therapeutic puzzle
They are discreet, but formidable. The so -called persistent cancer cells, with high metastatic potential, are responsible for around 70 % of the death -related deaths. Their particularity? They escape classic treatments and come back stronger. These cells, identified in particular by a strong expression of the CD44 protein on their surface, have a unique capacity: they actively internalize iron via this marker. This process feeds their aggressiveness and plasticity, making them capable of adapting to various tumor environments and surviving chemotherapy.
But this greed for iron is also their weakness. By accumulating this element, they paradoxically become more vulnerable to a cellular death mechanism called ferroptosis. This process, catalyzed by iron, leads to the degradation of lipids which form the internal membranes of the cell – a fragility that the researchers have been able to transform into therapeutic lever.
Strike at the heart of the most resistant cells
Led by the biomedicine laboratory of the Curie Institute (CNRS/Inserm), the team of chemist Raphaël Rodriguez developed a hybrid molecule called Fentomycin (Fento-1). Its design is based on a doubly targeted approach: first, it accumulates specifically in lysosomes – these cellular compartments rich in iron of CD44+ cells – thanks to a sequence capable of crossing the plasma membrane and penetrating the cell via an endocytosis mechanism. Then, it releases an active fragment which locally intensifies the reactivity of the iron.
The following is a lethal chain for the cell. Iron, reacted with hydrogen peroxide naturally present in lysosomes, produces extremely unstable oxygen radicals. These radicals tear the lysosomal membranes, then spread throughout the cell by oxidizing the lipids of other organelles, to cause generalized disorganization. The cell tries in vain to repair these breaches, but its inability to restore its membranes condemns its functioning.
 © CNRS/INSERM
© CNRS/INSERM
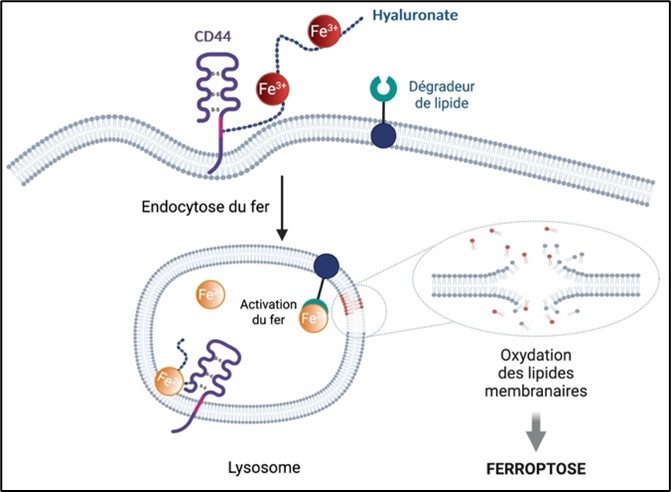
Iron enters cancer cells via the CD44 protein presents on their surface allowing it to acquire metastatic properties and tolerance to conventional treatments by epigenetic reprogramming which plays a key role in cellular adaptation. The activation of the lysosomal iron by a phospholipid degrading causes oxidation and the rupture of cell membranes, thus resulting in the death of the cell.
Fentomycin thus acts as a catalyst for ferroptosis, exploiting iron overload to reverse the advantage of the most dangerous metastatic cells.
Results to be confirmed in a clinical study
Beyond the theoretical elegance of mechanism, experimental results reinforce the credibility of this approach. After administration of fento1 in preclinical models of metastatic breast cancer, researchers observed a clear decrease in tumor growth. Even more striking: tests carried out on human biopsies – from pancreas and sarcomas – revealed a marked cytotoxicity of the molecule, even though these tumors often resist standard treatments.
By making the molecule visible thanks to its fluorescent property, the researchers were able to confirm its precise location in the lysosomes, thus validating the expected intracellular targeting.
This breakthrough opens an unprecedented therapeutic path: tackling the most stubborn cells by triggering in them an inner, controlled and fatal fire. If clinical studies remain necessary to demonstrate the effectiveness of fento1 in humans, the results obtained already place this molecule as a serious candidate to complete, or even strengthen, the arsenal against refractory cancers.