
Born with a rare and serious malformation, children with an esophageal malformation cannot eat normally and must resort to a probe and then complex surgeries. But researchers are offering an alternative: biological tissue manufactured in the laboratory to regain this vital function. A world first that revolutionizes the outlook for pediatric surgery.
A suspended start to life: when food becomes impossible
It all starts with a brutal diagnosis. At birth, some children suffer from esophageal atresia: their digestive tract is interrupted, preventing normal eating. A rare condition, but with immediate and serious consequences.
In the most severe cases, an entire portion of the esophagus is missing. Children cannot survive without surgery, but the opening is often too large to close immediately after birth. As a result, these infants typically need a feeding tube inserted directly into their stomach to ensure adequate nutrition while the medical team develops a treatment plan. Current surgical solutions are both complex and invasive. One method involves repositioning the stomach or intestine to close the opening, which involves two major procedures with significant short- and long-term complications, such as respiratory and gastrointestinal problems, as well as an unknown long-term risk of cancer.
Although many children have good outcomes, it is crucial to develop better options with less risk of complications for these babies. The scientific challenge is dizzying: design a tissue capable of replacing a portion of an organ, integrating into the body and functioning sustainably.
Making living things: the feat of bioengineered tissue
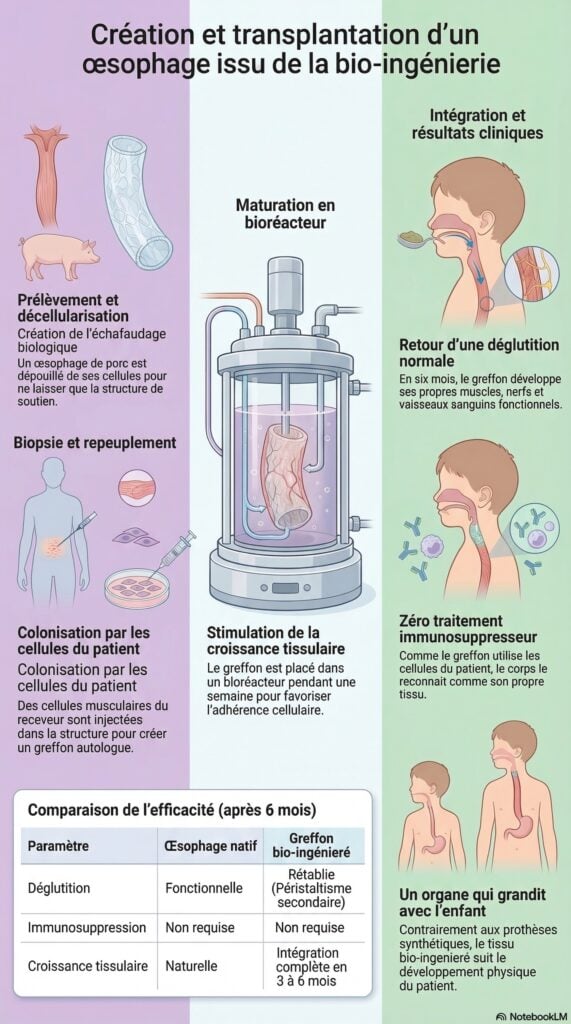
The first phase of this innovative technology involves the creation of a matrix serving as a tubular support for the new organ. Researchers use the esophagus of a donor pig, which is very similar to that of humans. Through a process called decellularization, the donor tissue is carefully cleared of all porcine cells, while retaining the underlying support structure.
Professor Paolo De Coppi, pediatric surgeon at the UCL Great Ormond Street Institute of Child Health, explains: “The esophagus is a very complex organ, lacking its own vascularization; it cannot therefore be transplanted in the classic sense of the term. To develop alternatives, it is essential to work with animal models that faithfully reproduce human anatomy and functioning. In this respect, the pig’s esophagus is very similar to that of humans..
Then, this structure is repopulated with muscle cells from the recipient pig, obtained by a small biopsy. These cells are multiplied in the laboratory before being injected directly into the matrix. The graft is then placed in a bioreactor, a special device that circulates essential growth fluids through the tissue for a week. During this time, the cells establish themselves, grow and adapt to their new environment. This entire process takes two months, which is consistent with current standard treatment for esophageal atresia.
Studies carried out on pigs have produced very promising results, paving the way for treatments for humans. All eight animals survived the critical 30 days following transplantation. After six months, the lab-grown grafts had developed functioning muscles, nerves and blood vessels. The transplanted esophagus could contract and move food forward like a natural esophagus. The transplanted animals were able to eat normally and their growth was healthy. Although some developed strictures, these were successfully treated endoscopically, as is commonly done in humans.
And the changes are observable at the cellular level. For the first time, this team was able to map the genes of the structure of the implanted tissue, showing that the genes activated in the new esophagus matched those of natural tissue. A progressive regeneration of normal esophageal structures was also observed, with the presence of a barrier layer, muscles, nerves and blood vessels necessary for the proper functioning of the esophagus. The bioengineered esophagus was found to be able to contract, producing movement and sufficient pressure to allow normal swallowing.
An alternative possibly available within 5 years
Ideally, if this technology is adapted for human use, matrices of varying sizes, derived from donor pigs, could be stored, ready to be expanded and customized for newborns or children of all ages and sizes, as needed. Biopsied cells could be taken from the child during feeding tube installation and integrated into the matrix as described in this research, creating a personalized graft that would grow with the child without the need for immunosuppressants. This “custom-made” tissue would then be surgically implanted to fill the missing part of the esophagus.
Dr Marco Pellegrini, principal investigator at UCL Great Ormond Street and co-leader of the study, is optimistic: “Our technology could make it possible to reconstruct a child’s esophagus from their own cells, taken during surgery anyway, and combined with a pre-designed matrix made from porcine tissue. Since the graft contains the child’s own muscle progenitor cells, it would be recognized as its own tissue. It could thus evolve with it over time, without risk of rejection and without requiring long-term immunosuppression..
“Thanks to the success of this research, we hope to be able to offer a bioengineered tissue alternative within five years to children who desperately need it.” testifies Professor Paolo De Coppi.
Between promise and caution: a revolution still under construction
While this advance gives rise to immense hope, let us remember that we are still in the early stages of this technology. Each case is unique, and long-term follow-up will be crucial to assess the durability and safety of these artificial tissues. This world first in animals nevertheless opens a gap. It could, ultimately, transform the management of numerous congenital malformations, but also, potentially, other pathologies requiring complex reconstructions.
Beyond technical prowess, a new way of thinking about medicine is emerging: medicine capable of manufacturing living things, tailor-made, to repair humans. And for the families concerned, this progress is not abstract. It embodies a new possibility, where there were sometimes only limited alternatives.
Between technological innovation, medical audacity and human fragility, this intervention marks a turning point. And in the face of these tormented family stories, every progress counts. Because beyond the figures and publications, there is a simple reality: allowing a child to eat, to grow, to live. This is perhaps where, ultimately, the scope of such progress can be measured.
 © Notebook LLM
© Notebook LLM